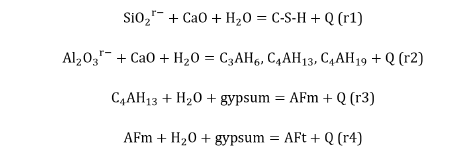Current Issue
Decarbonization in Cement Manufacturing-The Role of Calcined Clay for the Achievement of Environmental Neutrality by the Cement Industry: Its Reactive Alumina Content (Al2O3r–) or Key Chemical Parameter for Verifying Its Quality
Rafael Talero1, Carlos H. Arámburo Varela2, Rafael Talero3*
1PhD student, Salamanca University, Spain
2Retired, Cement Manufacturer, Cali, Colombia
3Retired, Eduardo Torroja Institute of Construction Science – CSIC – Madrid, Spain
Corresponding author: Rafael Talero , Retired, Eduardo Torroja Institute of Construction Science – CSIC – Madrid, Spain, Phone: 617 819 911, E-mail: [email protected]
Received Date: January 29, 2026 Published Date: April 29, 2026
Citation: Talero R, et al. (2026). Decarbonization in Cement Manufacturing- The Role of Calcined Clay for the Achievement of Environmental Neutrality by the Cement Industry: Its Reactive Alumina Content (Al2O3r–) or Key Chemical Parameter for Verifying Its Quality. Material Science. 8(1):43.
Copyright: Talero R, et al. © (2026).
ABSTRACT
The large-scale thermal activation of clays to produce highly reactive pozzolans is a critical technology for reducing CO₂ emissions in cement manufacturing and make it much more sustainable. This document establishes the scientific and technical principles for producing high-quality supplementary cementitious materials (SCMs) from thermally activated clays. The quality of these SCMs is primarily determined by their reactive silica content (SiO₂r⁻ (%)) and, most importantly, their reactive alumina content (Al₂O₃r⁻ (%)). We examine key manufacturing aspects, including pyro-processing and separate grinding technologies, and discuss the optimized use of these materials in novel blended cements. These cements exhibit superior sustainability, being more eco-efficient and environmentally friendly than conventional Portland cement. Furthermore, they demonstrate enhanced mechanical performance and durability, particularly showing improved resistance to chloride-induced corrosion of steel reinforcement, sulphate attack (with replacement amounts greater than 40-55%), seawater exposure, and alkali-silica reaction (ASR). This paper highlights that the reactive alumina content (Al₂O₃r⁻ (%)) is the key chemical parameter for verifying the quality and performance of activated clay” (AC) in sustainable cement applications. Because the higher this content, the greater the PC replacement amount that can be achieved, regardless of whether the kaolinite content of its matrix clay is greater or less than 40%. Because against the subsidiary generation of CO2, when decarbonating the limestone from the Portland clinker raw material in its kiln, in 1st phase, the subsidiary generation of H2O vapor from the dehydroxylation of the clay when it is thermally activated in its kiln is also taken into account, in such a way that, as long as higher the quality of the matrix clay, greater Al₂O₃r− (%) content will have activated and greater percentage of replacement will allow by OPC, which make the manufacture of the new OPC/AC blend cements much more sustainable: directly and indirectly.
Keywords: Calcined Clays, Thermal Activation, Dihydroxylation Process, Reactive Alumina, Pozzolanicity, Quality Control; Joint Grinding, Separate Grinding, Blending Station
INTRODUCTION
Currently, the cement industry is working on the search and use of new supplementary cementitious materials (SCMs) that allow the clinker/cement ratio to be reduced significantly to make its manufacturing more sustainable. Those traditionally used in Spain are GGBFS, natural pozzolans and fly ash, above all and very specially. In the case of the latter and given the obligation to reduce “Greenhouse Effect” gas emissions and the commitments acquired from COP21, the systematic and planned closure of coal-based electricity generation plants has already begun, so their availability will vary to be greatly affected and seriously compromised in the next future.
In Figure 1, the scarce availability of conventional SCMs can be observed in relation to the limestone and activatable clays. The availability of clays that can be thermally activated is quite large, which makes them the SCM with the greatest potential for the cement industry and even more so considering the drastic decrease in the supply of fly ash that is going to be produced shortly.
Figure 1: Availability of MCSs worldwide. Their variability depending on their chemical composition.
The need then arises to thermally activate the clays because they are also an MCS with very high pozzolanic reactivity where the cement manufacturer also has control of its production capacity and quality, in addition to its rapid verification, above all, being necessary, therefore and before of all, give clarity to the terms and differentiate "calcined clay" from "thermally activated clay", since the first term, which is the one that is appearing the most now in articles [1-4] and in other publications, also includes calcined clays from the ceramic and brick industry whose pozzolanic activity [5] is very low and slow [6-11] because it is mainly due to the cristobalite amount or reactive silica (SiO2r–), resulting from the thermal transformation of the matrix clay, along with mullite (3Al2O3.2SiO2), which is also formed at the same time and is very stable in strong acids and bases [12]. However, the second term does not include them because their pozzolanic activity is much greater and much faster due not only to the reactive silica (SiO2r– (%)) amount resulting from the thermal activation of the matrix clay but also, and above all, to the reactive alumina amount (Al2O3r– (%) or tetra and/or penta-coordinated Al [13]) that has also been formed at the same time for, which reason, it should be called “thermally activated clay” or simply “activated clay” (AC) but not “calcined clay”.
In this way, the cement sector can move towards greater sustainability through the industrial development of this new technology to produce CC in each of its factories. Since this production process can even have a very considerable reduction in CO2 emission levels: close to 70% compared to the manufacture of Portland clinker, depending on the quality of the clay, i.e., depending on its final Al2O3r– (%) content, especially and above all, after thermal activation. Without detriment either and, on the other hand, also a very important decrease in energy consumption because the thermal activation process of the clay requires lower temperatures than those of clinkering.
CLASSIFICATION OF POZZOLANIC ADDITIONS BY THEIR CHEMICAL CHARACTER
Classifying and cataloguing pozzolans based on their origin, natural or artificial, or by their total oxides content and, ultimately, based on their chemical composition [14], is insufficient and in no way significant for the characterization of their pozzolanic reactivity [14-16]. For this reason, R. Talero proposed a very different classification based on the results and conclusions of his investigations carried out with or without other authors 17-48], which is based on its reactive chemical nature as a result of its corresponding reactive silica, SiO2r (%) content [12,13,49], but, above all and very especially, of its reactive alumina, Al2O3r– (%) content [12,13,50], (tetra– and/or penta–coordinated aluminium, Al), to which, in the case of metakaolin, a metastable Al structure similar, in a certain way, to the crystalline phase of χ-alumina must be assigned [13]. In other words, based on the benefits that, under equal circumstances and conditions, each pozzolanic addition, natural or artificial, that forms part of cements and/or their derived products: concretes, mortars, pastes, grouts and premanufactured elements, may provide sooner or later depending on the physical state of the pozzolan, amorphous or vitreous, its reactive chemical composition as well as the shape and average size of its particles.
The chemical reactions in which the hydraulic factors: SiO2r– and Al2O3r–, of the pozzolans are involved are detailed below when they chemically react with slaked lime in an aqueous medium and at room temperature, at faster, logically, with the portlandite of the hydrated fraction of Portland cement with which they have been mixed:
In this sense, it is of utmost importance to be able to know, therefore, the chemical character of the pozzolan through the determination of its hydraulic factors: SiO2r– content (%) [49] and Al2O3r– content (%) [50]. Since, depending on their different contents and the relationship between both, the chemical character of the pozzolan will also be different, being therefore and equally its influence very different in all the properties, performances and behaviours of the Portland cement base materials of which it forms part. From:
- the fresh state of your PC/AC pastes: rheological and thixotropic behaviour [47,48], to
- the freshly set and hardened state: calorimetric behaviour [43,46], and to
- the fully hardened or completely hardened state:
- mechanical strength [17,32,37-39], and
- durability: behaviour against attack:
- from sulphates [17-34] and chlorides [35-40], separately and exclusively, or simultaneously: seawater attack, and
- from all other natural aggressive chemical attacks, they must face: AAR (ASR or ACR) and carbonation, whose chemical basis is the same but its consequences on the concrete are completely opposite, such that its effectiveness on the AAR must be preserved by simply liming the concrete element,
which can also be influenced by for better or worse the shape and average size of its particles, in the case of pozzolans which are of siliceous nature and of silicic chemical character type: silica fume [7,29-31,39-43,46] and diatoms [17,43,41-43], respectively, and in all other natural and artificial ones that are siliceous and aluminous in nature [14-16] and alumic in chemical character [17] or a random mixture of both extreme chemical characters. But with supremacy always of one chemical character over the other without having to infer that the percentage content of the hydraulic factor that supports and justifies said supremacy must be greater necessarily and obligatorily. Because, in equal circumstances and with the same everything else (including the average size and the distribution of its particles), the chemical reaction speed of each one (of SiO2r– and of Al2O3r–) with the portlandite and/or with the slaked lime in an aqueous medium and at room temperature, with the sulphates and/or chlorides being present or not, is quite different, although that of the Al2O3r– always holding priority, supremacy or preponderance [17,29,37,39-43].
THE CLAYS. ITS THERMAL ACTIVATION TO PROVIDE THEM PRONOUNCED POZZOLANIC CHARACTERISTICS
The term "clay" refers both to a group of phyllosilicates and to a granulometric division of detrital rocks. This term also loosely designates a sediment or rock made up largely of clay minerals [51].
From a granulometric point of view, clay is any fraction less than 1/125 mm (≈ 4μm) of a detrital rock, regardless of its composition. Although there is no precise size limit for clay minerals, most do not exceed 2 μm.
Clay minerals belong to the group of phyllosilicates and occur in very small crystals, micrometric in size and with a laminar or fibrous habit. From a chemical point of view, they are hydrated aluminosilicates, originating from the chemical alteration of igneous and metamorphic rocks and incorporated into sedimentary rocks as detrital minerals. The rest are formed in certain sedimentary and paedogenetic environments or during diagenesis. During the sedimentary cycle, clay minerals can undergo degradation transformations (structural deformation, ion depletion) or the opposite process (ion fixation, regeneration of degraded structures). In turn, the igneous and metamorphic rocks from which they come are the most abundant minerals in the earth's crust –silicates– in which they occupy 60 to 65% of its total volume, with a crystalline structure that allows a great capacity for exchange and retention of water.
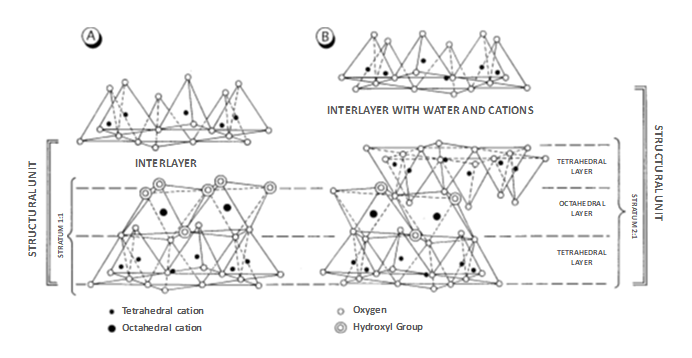
Said crystalline structure is essentially made up of two types of layers: tetrahedral and octahedral [52]. The tetrahedral layer has the Si2O5 group as the basic unit, with silicon in tetrahedral coordination and three oxygens from each tetrahedron shared with the adjacent ones, forming a hexagonal structure (Figure 2). A part of the silicon atoms can be replaced by aluminium atoms and, occasionally, by Fe (III).
Instead, the octahedral layer is constituted by a cation, generally Al, Mg, Fe (II) or Fe (III), in octahedral coordination with oxygen or hydroxyl ions. The smallest structural unit of the octahedral layer consists of three octahedrons and depending on the degree of occupancy of the octahedral positions, the layers can be dioctahedral or trioctahedral. Dioctahedral minerals are those in which only two of the three octahedrons of the structural unit have a cation in the centre. When all the octahedral positions are occupied, the minerals are called trioctahedral, and in this case the cations occupying the octahedral positions are divalent (Mg, Fe (II)), while in dioctahedral minerals the octahedral positions are divalent. occupied by trivalent cations (Al, Fe (III)).
Figure 2: Structure of clay minerals. A) Stratum of the 1:1 type. B) Stratum of the type 2:1 [51].
The stacking of the layers and the substitutions of the ions determine different types of clay minerals. Depending on the stacking of the layers, they are divided into two large groups: those made up of a tetrahedral layer and another octahedral layer, which share oxygen atoms (type 1:1) and those made up of two tetrahedral layers separated by an octahedral layer (type 2 :1). Each of these units is called a "stratum." The space between two layers is called the "interlaminar space" (Figure 2). When the layers are electrically neutral, that is, when the structural cations are balanced by oxygen or hydroxyls, the interlaminar spaces are empty. However, the layers often have a negative charge due to cation substitutions, and in these cases, they are held together by cations and hydrated cations that balance this charge. The most common interlaminar cations are potassium, magnesium, calcium, and sodium. Sometimes, the occupation of the interlaminar space is structured in an additional octahedral layer, called “brucitic layer”, and gives rise to 2:1:1 type clay. The set of a stratum plus the interlaminar layer constitute a structural unit that serves as the basis for establishing the classification of clays [53] that will be seen later. Other criteria that are considered, although less, are the chemical composition and the geometry of the superimposition of the strata. Here often, clay minerals are not made up of just one type of layer but rather have stacks of two (rarely three) different types. These structures are called “interstratified”. The stacks can be regular or not. When they are regular, the minerals receive specific names (corrensite, rectorite, etc.). However, the most frequent case is that of irregular stacking and then, the minerals adopt the names of their components. The most common interbedded ones are illite-smectite and chlorite-smectite. Interstratification is possible because of the weak chemical and structural bonds between the strata.
Other criteria that are considered, although less, are the chemical composition and the geometry of the superimposition of the strata. Here often, clay minerals are not made up of just one type of layer but rather have stacks of two (rarely three) different types. These structures are called “interstratified”. The stacks can be regular or not. When they are regular, the minerals receive specific names (corrensite, rectorite, etc.). However, the most frequent case is that of irregular stacking and then, the minerals adopt the names of their components. The most common interbedded ones are illite-smectite and chlorite-smectite. Interstratification is possible because of the weak chemical and structural bonds between the strata.
Generally, the overall chemical analysis of a clay sample provides very little information, since most of them contain various clay minerals along with other accompanying minerals. The practical impossibility of separating pure phases means that global chemical analysis has a relative and highly subjective value and, furthermore, its application is reduced to mono-mineral samples or to the resolution of some specific problems.
Finally, another typical chemical parameter of clays to consider is their “cation exchange capacity” (CEC). It is used to determine the number of exchangeable ions in the clay that can be fixed in its crystalline structure. Both in the interlaminar spaces and on the surface of the grains [52].
Clays are therefore made up of sedimentary rocks formed, as mentioned before, by phyllosilicates of the so-called «clay mineral group», which includes, in turn, the following groups [53]:
- Kandite group (main mineral: kaolinite).
- Group of illite (main mineral: illite).
- Group of smectites (main mineral: montmorillonite).
- Vermiculite group (main mineral: vermiculite).
- Palygorskite group (main mineral: palygorskite).
All clay minerals have a laminar habit, except those of this last group, which present fibrous morphology. Of the five groups, the most likely to be activated thermally are those of the first three. The clays contain a maximum of 60% phyllosilicates. Its remaining components are detrital minerals (quartz, iron oxides, feldspars, etc.) or neoformation sedimentary minerals (carbonates and sulphates). Among iron oxides you can find hematite, among carbonates, calcite, and dolomite, and among the sulphates, gypsum. Likewise, they can also contain zeolites, sulphides, etc., as well as amorphous or slightly crystalline components, such as opal, imogolite or allophane, while within the thickest fraction fossils, organic matter and rock fragments can be found [54].
Sedimentary clays can come from two different formation processes. They are considered as original:
- Primary (residual clays): those formed by the alteration of a "mother" rock and remain in the place of their formation. This is the case of kaolin, originated by the hydrolysis of pelitic and quartz-feldspathic rocks, which will be seen in more detail in the following paragraph.
- Secondary (transport clays): those other clays formed by transport and subsequent sedimentation of detrital elements. They are more frequent than the previous ones and have much larger and more powerful formations and may present a very different appearance depending on their different location.
Pelitic rocks are characterized by the presence of highly aluminium minerals. The metamorphic varieties derive from clayey materials and include slates, schists, pelitic gneisses and cornubianites. The quartz-feldspathic rocks, greywackes, arkose and acid igneous rocks, are transformed, on the other hand, into gneisses or corneas.
Most of the pelitic rocks are adequately described in the KFMASH chemical system, whose components are: K2O, FeO, MgO, Al2O3, SiO2 and H2O [55]: In addition, it may contain more important minority components: Fe2O3; TiO2, MnO, CaO, Na2O and C, components that can generate particular subsystems capable of forming new minerals or changing known mineral stabilities [56-58]. In relation to pelites, quartz-feldspathic rocks generally contain lower proportions of Al2O3, FeO, and MgO, and higher proportions of K2O, Na2O, and CaO. The most suitable system for its study is in this case, the CKNASH [58].
The metamorphic minerals of the pelitic rocks correspond, mainly, to the mineral phases of the AFM triangular diagram [59]: pyrophyllite, andalusite, sillimanite, kyanite, staurolite, chloritoid, lambskin, garnet, hypersthene, chlorite, biotite, as well as quartz and muscovite. or potassium feldspar. The metamorphic mineralogy of the quartz-feldspathic rocks is expressed in the AKN: Al2O3-KAlO2.NaAlO2 and ACN: Al2O3.CaAl2 diagrams. O4.NaAlO2 [59,60]. The mineral phases that are described in these diagrams are: pyrophyllite, andalusite, sillimanite, kyanite, albite, anorthite, jadeite, potassium feldspar, lawsonite, daisy, paragonite and muscovite.
Pelitic rocks are good indicators of metamorphism because they are highly sensitive to changes in pressure and temperature. This characteristic is given by the great chemical and mineralogical variety and by the fine grain size that they generally present at low and medium grade. In contrast, quartz-feldspathic rocks are more refractory. In this case, the characteristic mineral associations appear only under high grade and/or high-pressure conditions.
Finally, among the hydrothermal alterations in granites s.s. of epizonal or mesozonal emplacement is the "kaolinization" which is an alteration characterized by the replacement of micas and feldspars by kaolinite and the dissolution of quartz, creating almost monomineral masses of white colour. Geochemically, it represents an extreme alteration that only occurs in low-temperature systems (< 200 °C) with circulation of very acid fluids. Examples of kaolinitinized zones are those found in Cornualles [61]; in Spain, Penouta or Golpejas [62,63]. Like this is "argilization", a process that involves the formation of clays (kaolinite and montmorillonite) at the expense of feldspars and micas.
Macroscopically, the clayey rocks are soft, earthy, finely divided, and powdery, being able to fall apart by hand and having an odorous and soft touch, being made up of crystalline aggregates of hydrated silicates and aluminates, as mentioned before. They are highly variable in colour: grey, green, red, brown (these last two colours are usually justified by the lesser or greater presence of hematite (Fe2O3)), etc. The black varieties are very rich in carbonaceous substances, although they tend to be white when they are very pure.
Both kaolinite, montmorillonite and illite, subjected to adequate heating, can be activated because of a dihydroxylation process that suffers or loss of OH– groups from their crystal lattice, through the following chemical reaction:
+ Q―OH– + ―OH– ――> H2O↑ + O2–
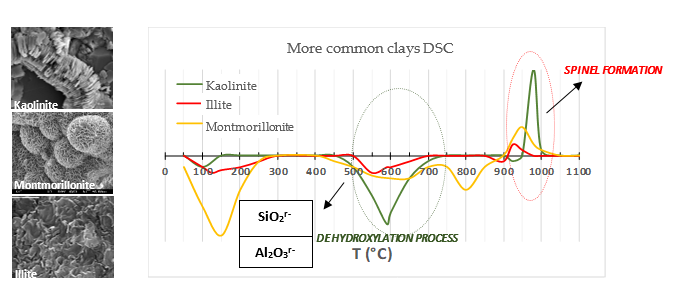
The optimum temperature by which this purpose is achieved usually ranges between 600 °C and 800 °C, depending on the clay mineral composition of the clay. In short, the thermal decomposition of clay begins at 120 °C with the loss of moisture (hygroscopic, colloidal and hydration water, and the physically adsorbed, or absorbed in pores of the clay material). Afterwards, and as the temperature of the process increases, the hydroxyl groups that are part of the crystalline network of the clay begin to separate from it (dihydroxylation stage [64]), due to the increase in vibration energy that is conferred on them, reaching the value or degree of thermal agitation sufficient to be able to unite with a nearby proton and form a water molecule that goes into the atmosphere to finally separate of the crystalline structure, leaving it, moreover, in an amorphous state [13,50] that is not vitreous like that of fly ash [37,38,65,66] and silica fume [30,31,39-41,46]. At temperatures above 920 °C the activated clay becomes very unstable and the formation of spinel, pseudo-mullite or pre-mullite and even mullite is possible [12] (continue in paragraph 3 of Section 5). Figure 3 shows the thermal behaviour of the most common clays [64]. The temperature values mentioned here correspond to kaolinitic clays.
Figure 3: Thermal behaviour of the most common clays and its consequences for their resulting final pozzolanicity at each temperature [64].
Through the process of thermal activation of the clays, an artificial pozzolan with an aluminic chemical character is being produced, according to R. Talero [17-29,31-48], since by releasing the chemically combined water from the clay into the atmosphere, it is acting on the coordination index of aluminium which was 6 [12]being, after this thermal process, in optimal conditions, in coordination 4 and/or 5 [13] to react chemically and very quickly with the portlandite of the Portland cement hydration. Well, not in vain and, as it has just been said, its physical state is amorphous which justifies, on the other hand, that the pozzolanic reaction of the latter must be and is in fact, all other things being equal, much slower, necessarily. For this reason, and although kaolinitic clays have the highest aluminium content, there should not be any exhaustive restrictions a priori on which clay can be used to produce an artificial pozzolan and which cannot. Its suitability should be evaluated based on its SiO₂r⁻ content (%) but, chiefly, on its Al2O3r– (%) content, which can be generated after thermal activation and in anaerobic environment consistent with its reddish colour (more or less). This allows its pozzolanic activity to begin to be significant, as well as very rapid and early, and its grey colour in the case of red clays. For this last content (Al2O3r– (%)) of the AC produced to be the most significant of the two, it must be greater than 8% [17,32-34,39-4,43,47] and it must also be a random mixture of tetra– and penta–coordinated Al [13] which is the most common. Because if said mixture is not mixture but tetra–coordinated Al only, said limit may be somewhat lower, but, on the other hand, if it is only penta–coordinated Al it must be higher, logically, to originate and develop the same pozzolanic activity of this other origin as said random mixture. Which, in any case and without a doubt, quite justifies the fact that the mineralogical limitations and restrictions established by some researchers [67-69], that the selected clay material contain at least 40% kaolin for its thermal activation to be of environmental, technical (clays with a kaolin content greater than 40% achieve a mechanical strength greater than CEM I after 7 days or a little more) and economic interest, do not make much sense. And it makes even less sense to ascribe to metakaolin any precise and concrete chemical formula, whatever it may be, because, according to what has just been said about Al2O3r–, it does not have one and never will have one, as it is totally impossible. Whoever ascribes it is merely concocting or speculating. But, in addition, this minimum kaolin content of 40% contrasts quite significantly with
- On the one hand, at the 15th ICCC [70,71] Irassar et al. showed that a purely illite clay, thermally activated at suitable temperature, can be used to manufacture OPC/CC cements, although with addition of limestone filler (LF), such as LC3 [68,69] in order to mitigate, as far as possible and among other things, the notable and significant Heat Synergic Effect (HSE) that would be generated [45,46], due to the stimulation of hydration that the Al2O3r– (%) content of the AC would produce to the C3A (%) content of the OPC fraction with which it was mixed, instead, its corresponding SiO₂r⁻ (%) content does not. This stimulation of hydration occurs through:
1.1. Direct pathway [72-74],
1.2. non-direct pathway [75], and
1.3. indirect pathway mostly and above all [31-35,46-48], the latter being, in turn, a result of the very high, early and rapid pozzolanic activity that generates and develops the considerable Al2O3r– (%) content mentioned, from the beginning of hydration, which, due to its very high amount, overlaps and masks the first two, despite the fact that the authors had previously evaluated the pozzolanic activity of different activated clays (5 kaolins and 2 bentonites) [76]). Which would inevitably translate, among other things, once again, in microcracking and cracking of the newly set and hardened paste, due to plastic shrinkage [77], chemical shrinkage [78-83], autogenous shrinkage [83], hydraulic shrinkage [82], initial thermal shrinkage [82,84], drying shrinkage [83,85], and carbonation shrinkage [86]. Hence, the addition of limestone filler (LF) is highly appropriate to mitigate these effects given that, due to its geochemical origin, its intimate texture is granular and very particulate [72-75].
2. On the other hand, manufacturers of ceramic construction products are also very interested in them, both technically and economically. This will make it quite difficult for cement manufacturers to use them, because the profitability of cement manufacturing lies in the very low economic cost of its raw materials, and the economic cost of cement is often highly conditioned and regulated by political and social issues.
Therefore, and in accordance with such a notorious contradiction, the most reasonable and logical approach is to base the acceptance or rejection of the matrix clay for the manufacture of Portland-based blend cements or PC/AC type, with or without limestone filler (LF), on its SiO₂r⁻ (%) [49] content, but most of all, on its Al2O3r– (%) [50] content, after having been thermally activated at an appropriate temperature and aerobic environment. Because both contents, but especially the last one, also depend on:
- On the one hand, that the estimated maximum possible replacement of AC by PC is much more accurate than that estimated with the kaolin (%) content of its matrix clay, and
- On the other hand, its pozzolanic activity, which justifies its subsequent behaviour in: pozzolanicity test or Frattini test [13,15,87], water amount for normal consistency paste, setting times, volume stability, Strength Activity Index (SAI) test [88-90] and durability, and in all other standardized or non-standard tests and behaviours in real-life construction sites.
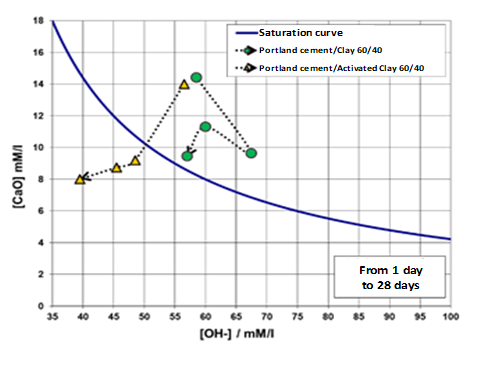
Finally, Figure 4 shows the pozzolanic activity of a kaolinitic clay, determined by the EN 196-5 [5] standard, before and after its thermal activation.
Figure 4: Increase in the pozzolanic properties of the clay after its thermal activation. Frattini test [5]. Results. Ages: 1, 8, 15 and 28 days. Saturation curve of Ca (OH)2.
PYRO-PROCESS TECHNOLOGIES
First, the clay must go through the drying, thermal activation and cooling processes. In addition, and in the case that the clay to be activated has a high iron content (Fe2O3 greater than 4%), it is important to guarantee the colour change of the clay to grey during the process of its thermal activation to favour its mixing and homogenization of colour with that of the Portland cement, at the same time, pozzolanic characteristics are generated that it did not have before as raw clay.
The main parameters to obtain thermally activated clay and guarantee its colour change are the precise control of the adequate calcination temperature and the oxygen concentration in the gases in the drying, thermal activation and cooling equipment.
The technology used for the combustion system of the drying and thermal activation processes allows operation with solid fuel, ensuring the stability of the flame even in a process with lower temperatures (less than 900 °C).
The drying and thermal activation of the clays can be carried out using rotary kilns or with “flash technology”. In the case of the use of rotary kilns, it is possible to reuse existing kilns in cement factories that are already out of service or not yet, to adapt them, in any case, to their new condition of adequate heating of the clays for its thermal activation.
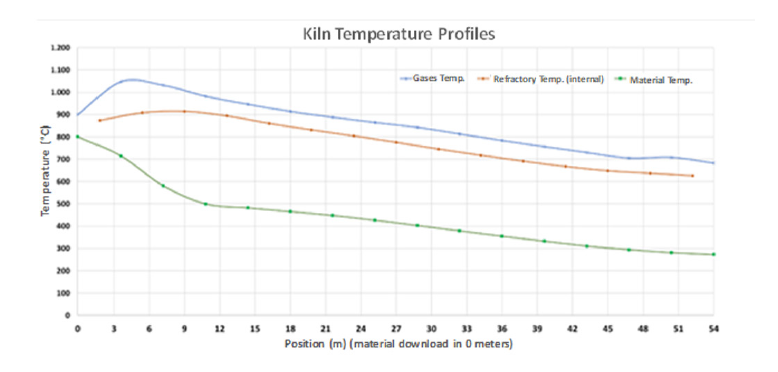
Figure 4 shows the temperature profiles of the gases; the bed of clayey material and the refractory lining of a mathematical model developed for the simulation of rotary kilns for thermal activation of clays. In this case, the clayey material fed into the kiln has already been previously dried at ≈ 250 °C.
Flash technology is based on the dragging of small solid particles by a concurrent flow of hot gases, which makes it possible to obtain high heat and mass transfer coefficients in more compact equipment. This technology can be used for the drying or activation process. In the case of applying a flash calciner, the process must be staggered in several stages in a cyclone tower to guarantee the adequate residence time of the clayey material in the temperature range of 750 to 850 °C or "transit time", necessary for thermal activation to occur.
The experience in rotary kilns at an industrial level is extensive in Brazil with kilns of up to 1.100 Ton/day and in Colombia with a furnace with capacity of 1.500 Ton/day. Its operation is relatively simple and easy for control room operators to understand. Its operation control, in terms of adjusting the activation temperatures according to the hourly quality control variables, is relatively easy to use, even though the short activation range in terms of temperature makes this control very demanding to be able to obtain AC with high pozzolanic activity. Therefore, it is important to say that the experience of thermal activation of clays with rotary kilns has already been going on for several decades and, today, there is no flash calciner activation of such an important industrial size.
Finally, and after having achieved the thermal activation of the clayey material, it needs to be cooled. In this last stage and in the case of clays with high Fe2O3 (%) content, it is also important to control the atmosphere to prevent the material at high temperatures from encountering high air flows to ensure that the grey colour obtained in the previous heating stages is maintained and is not lost by oxidation of the resulting new thermally activated material. A technology that is perfectly suited to these two objectives (cooling the material and maintaining its grey colour) is the rotary cooler.
Process Control and Verification Variables
One of the aspects that acquires extraordinary importance in the process of thermal clays activation is how to ensure their quality after having activated them because there is still no analytical method or mechanical strength test that determines the pozzolanic activity immediately after the activated clay (AC) has left the rotary kiln or flash calciner and has cooled to room temperature. Therefore, whether the thermal activation of the clay has been correct or not, it must necessarily be determined directly to, in the latter case, adjust the thermal activation process of the kiln (the calcination temperature and the transit speed of the raw clay) quickly so that it has the highest possible contents of SiO2r– (%) [49] and Al2O3r– (%) very especially and above all [50], after having been activated as it should. Which, without a doubt, will translate into greater pozzolanic activity, as stated previously.
During this stage and for the same verification reason, it is highly recommended to also determine, after this heating process, parameters such as the “Loss of Ignition” (LOI) [14-16,87-90] of the properly calcined material obtained and SAI [14-16,88-90]. Without prejudice either and, on the other hand, having determined residual water by LECO instrumental technique [91] which is much more precise and meaningful, and, if possible, the percentage of clay minerals that constituted the matrix clay, before its thermal activation. This will allow to correlate the results of all the analyses and verification tests [5,50] with the latter, for which it is also of utmost importance, in this previous stage, to have previously determined for each batch of clay to be thermally activated, its total contents of SiO2 (%), Al2O3 (%) and Fe2O3 (%) in order to, through their comparison, estimate with precision the particular temperature of greatest activation of each type and batch of clay in the deposit, the aerobic or anaerobic medium necessary and its transit speed in the oven because they are the ones that must confer the highest content of Al2O3r– (%) to its matrix clay. Which, in turn and once again, will allow us to estimate, with a fair degree of accuracy, the lowest possible clinker/cement ratio.
In this sense, the activation temperature at which the highest Al2O3r– (%) content [50] in the AC is obtained will be the optimal temperature. For this reason and in this way, the upper and lower activation ranges can also be determined, which indicate that if said optimal thermal activation temperature is exceeded to a certain extent, recrystallization of the initial totally amorphous structure of the AC obtained by having been sintered and pelletized as particles of any ceramic brick, also adhering as crusts to the walls of the furnace refractory, making transit through it difficult and thus losing its activation level and, therefore, pozzolanic activity [1-4,8-11]. But if, on the contrary, the temperature is not exceeded, but is lower than the optimum, sufficient, or necessary dihydroxylation is not achieved, and then its pozzolanic activity is also very poor because there are still traces of clay that are not thermally activated, which is not good for cement [14,15, 87,88]. Either kaolinite and/or illite and/or montmorillonite or a random mixture of all three or just two of them. Which, without a doubt, will and does translate, in fact, into a lower degree of Portland clinker replacement in the cement to be designed, dosed, and finally manufactured and produced. Be it the type that is among the following possible regulated ones [14,15] that can be manufactured with it, namely: CEM type II/A-Q, II/B-Q, II/A-M, II/B-M, IV/A, IV /B, V/A, V/B and C-M [92], and may also have the characteristic of protecting the steel reinforcements of their corresponding reinforced concrete from chloride attack [35-42], but only when the AC exclusively replaces Portland cement CEM type I, but not, however, they have the sulphate-resistance characteristic [17-29]. Unless the replacement of AC by any Portland clinker is even greater than 40%, 50% and even 55% or even more the higher its Al₂O₃r− (%) content [31-34] in order to provide this characteristic, but this time based, rather, on the chemical basis of sulphate-resistant "over-sulphated" cements or SR-GGBF/OPC cements [93,94] precisely and equivalent cements on a chemical basis [17-29] for which, as will be remembered, the resulting mixture with its setting regulator (natural gypsum stone and/or soluble anhydrite, even better if possible) will have to have from 5 to 12% sulphur trioxide (SO3) [17-29,86,94].Therefore, for this purpose, it is worth remembering here the 2nd conclusion of reference [19] which reads as follows: “The 15.05% of gypsum (= 7.0% SO3) added to each Portland cement with metakaolin (MK = AC that also contained ≈ 50% of quartz filler as impurity), tested according to the ASTM C452-68 method [95], did not act as aggressive but rather as a setting regulator”. Without forgetting, on the other hand, the possibility of also manufacturing hydraulic binders for roads that harden quickly [96] rather than slowly [97] if the Al2O3r– (%) content of the AC that constitutes them is considerable and, quite the opposite, if it is not.
Finally, with all this information obtained through the analyses and verification tests of the quality of the AC produced, which must necessarily be carried out at the laboratory level, it is possible to clearly identify and determine the ranges in which these variables move from according to the highest pozzolanic activity to be achieved, since the aforementioned analyses and tests must also be used in the quality control and verification of the AC produced on an industrial scale in the cement factory and/or concrete Plant.
Figure 5: T profiles of the gases, the bed of clayey material and the refractory.
JOINT GRINDING
The most common operation in cement factories is the joint grinding in a single mill of Portland clinker, its setting regulator (natural gypsum stone) and the active and/or non-active mineral additions that are incorporated in each case (SCMs). where the reduction in particle size of the Portland clinker, gypsum and SCMs occurs. For this reason, it is very important to know the hardness index of the different materials to be ground, their humidity, their proportions, and feed granulometry, in order, with all this information, to design the load of grinding bodies that each grinding chamber must carry. also, in accordance with the typology and physical and mechanical quality of the cement to be produced.
The AC has a very high fineness. It could be said that 85% of its mass passes through the 1 mm mesh sieve, although this value will depend on the type of clay, its mineralogical composition, and its quartz content. As an example, it can be mentioned that the Bond hardness index for limestone can range from 10 to 13 Kwh/Ton, for AC from 13 to 15 Kwh/on and for Portland clinker between 16 to 18 Kwh/Ton. The ranges may vary depending on its mineralogical composition, quartz content, origin, etc.
In some cases, the quartz content in the raw clayey material fed to the kiln can vary between 25 and 50%. This factor must be considered, therefore, in a joint grinding. The AC has an intermediate hardness, although it is closer to that of limestone, having a very fine feed granulometry, so it will be much easier to grind than limestone, leaving the first chamber of the mill quite empty. To obtain a performance of the same order of magnitude as traditional cements, it will surely be possible to work with the specifications of a higher retained material on the No. 325 sieve, although its Blaine fineness will also be greater. As an example, for a “General Use” type cement [98], with an expected performance of ≈ 26 MPa at 28 days, it will be possible to work with a retainer on the No. 325 sieve between 4 and 7% and a Blaine fineness. between 4500 to 5500 cm2/g.
In addition, quartz, although it is hard to grind, is not usually very large, so in this type of grinding it can play the role of "grinding additive", by also producing a cleaning effect on the balls and the emplacement. of the mill, helping the grinding, thus being able to reduce or avoid the chemical additive to improve the grinding itself. On the other hand, and due to the very low granulometry of AC, the possibility of feeding it directly to the separator can also be studied. The physical aspects discussed here gain strength for replacement levels above 12%, but, however, with a low diet, your changes will not be very noticeable. And as far as the pozzolanic activity is concerned, it will become important with replacement levels higher than 8% depending on its Al2O3r– (%) content.
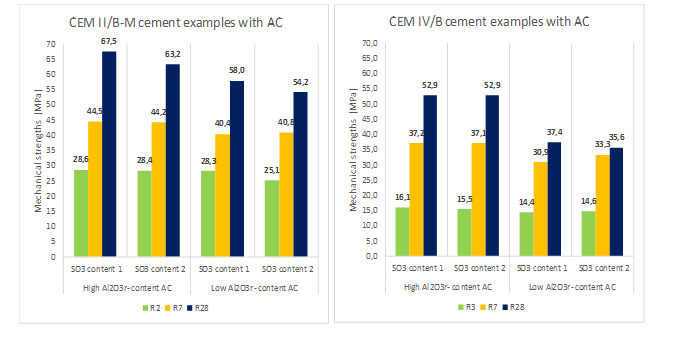
Regarding the dosage of the cements with this pozzolanic addition, the optimum ratio between all the components, Portland clinker, AC, other MCSs and gypsum, will depend on multiple factors. Therefore, each dosage must be studied and analysed separately and exclusively, based on the following premises or conditions: the mineralogical composition of the Portland clinker, the reactivity of the AC produced (its SiO2r– (%) content and Al2O3r– (%) content especially and above all) and the optimal SO3 content [17,19], among others. As an example, Figure 6 shows the mechanical performance obtained with two different types of cements in dosage: Portland clinker, AC, and limestone filler, and with the same grinding fineness, respectively. In this test, two AC with different Al2O3r– (%) content and different proportion of gypsum added as setting regulator were used.
Finally, it is important to also consider in the transport systems, hoppers, and dispensers, the very low granulometry of the CC and its rheological behaviour being so dry, because avalanches can occur in the hoppers and the control in the dosage can be somewhat difficult.
Figure 6: Performance obtained in the dosage of different cements according to the EN 197-1:2011 Standard [14].
SEPARATE GRINDING AND BLENDING STATION
Undoubtedly, the best grinding option to manufacture cements with several MCSs is separately. From the point of view of the grinding of the particles, the ideal is to grind materials of similar hardness because this ensures a more controlled and efficient grinding. In a co-grinding, the grinding is conducted by the hardest material, and this will define the retained on the No. 325 sieve and the Blaine fineness, therefore, the softer materials will be "over-ground" and these will affect the final granulometric distribution. of the product. Logically, the viability of this system will depend on the availability of equipment: two mills and their production capacities.
Separate grinding has great advantages: it specializes the mills and therefore their grinding efficiency improves in terms of production and energy consumed and, most importantly, it eliminates setup times when switching from one type of cement to another (in terms of AC content). Since this preparation time for grinding a cement with a low AC content to another with a considerably higher content, it is a very important economic saving that occurs in grinding separately and that must be taken into account, since, Generally, these preparation times mean the production of a higher quality cement that is deposited in the silos of a cement with lower performance, until quality compliance is ensured, which undoubtedly translates into operational inefficiencies. Another great advantage in specializing the mills is that the operators of the room will not have to keep changing the conditions of the mills and their driving will be easier to perform and carry out.
However, another very important possibility is also having two “intermediate products”: the first, a “base cement” and the second, a “pozzolanic mix” cement that contains the materials that are easier to grind. The base cement can be made up of Portland clinker and setting regulator only and/or some additional SCM, depending on the types of cement that are intended to be produced. The pozzolanic mix will be made up, instead, of the AC, the rest of the SCMs and the appropriate amount of setting regulator according to its composition. The mixing percentages of each of the intermediate products will depend on the quality of its components and the qualities or types of cement to be produced.
Grinding the pozzolanic mix separately allows better control in terms of Blaine fineness and the retained material to look for, since, in this case, AC materials are easier to grind than Portland clinker. Depending on the materials to be ground in this pozzolanic mix, a "coarser" grind can be sought in terms of retained on the No. 325 sieve, it could well be in a range between 10 to 15% and the Blaine fineness would be a result. This physical aspect will be of great importance in the final fineness and in the conformation of the pore system in the final structure of the cement paste produced, which will certainly be more compact than that achieved in joint grinding. Which, without any doubt, will also result in greater mechanical resistance and durability of the cements to be produced in separate grindings with Blending stations.
Finally, and as it has been seen, separate grinding requires setting up Blending stations, simple in their operation and design and at the same time very demanding in terms of the Dosage Systems to be used, because herein lies the success of this. A Blending station is equivalent to having a new mill with high capacity and very high efficiency. In addition, it does not need preparation times and only introduces the corresponding mixture of the two intermediate products mentioned: the "base cement" and the "pozzolanic mix". The gain in terms of flexibility of the operation, dispatch logistics and customer service is unquestionable. For this technology it is essential to have a high-quality equipment for dosing and mixing. Its design capacity will be determined by shipping and storage conditions.
Once the multiple and varied purely physical aspects have been exposed that justify the grinding separately and exclusively or not, of the AC (plus some other active mineral additions –FA, NP, SF, GGBFS– and non-active or silica or limestone filler mostly and above all), on the one hand, and, on the other, the manufactured Portland clinker (plus its optimal amount of setting regulator [17,19]), followed by a Blending station, which are justified by their very different degree of hardness (physical parameter), it is now appropriate to also expose and describe the multiple and varied purely chemical aspects that occur between the two, once dosed and mixed for each constructive use in the Blending station and their joint hydration has begun, which also justify grinding them separately as will be seen below.
And when this joint hydration intervenes in acid><base<>solid><liquid chemical reactions, provided that the average size of the natural or artificial pozzolan solid particle (nanometric and micrometric at most in the case of AC), on the one hand, is smaller than that of the OPC, on the other hand, its joint hydration will occur to a greater extent and at a higher speed, but always initiated and promoted by the reactive fraction or hydraulic factors of the pozzolan (SiO2r– and Al2O3r– (%)). Especially if:
- On the one hand, its Al2O3r– (%) content is considerable [32-34,43-48] (as is that of most AC) and that of C3A (%) content especially of the OPC with which it was mixed, is as well [17-29][37-48]; because it is Al2O3r– (%) specifically (but not SiO2r– (%)) that really initiates, promotes and facilitates it since, under the same circumstances and everything else being equal, its pozzolanic reaction rate with portlandite is considerably greater than that of SiO2r– (%) (with sulphates [17-29,31-34] and/or chlorides also present [35-42] or without being present because it is only hydrated with water initially saturated with portlandite [37-39,43-48]); to the point that it rather seems that the SiO2r– (%) is inhibited and, not only is it not inhibited, but also, with the course of its corresponding pozzolanic reaction with the portlandite only, it ends up opposing the hydration of the rest of the OPC hydraulic components with which its pozzolan was mixed [7,17,29-31], and to such an extent it ends up opposing that, in an adequate amount to its aluminate Phase content (C3A (%) + C4AF (%)), it manages to prevent the attack of sulphates to its OPC concrete [17,29-31] and that of chlorides to its reinforcements [37,38], and with adequate water curing before [17,29-31,64] even more prevents, and,
- On the other, the physical state of both hydraulic factors is amorphous for AC and vitreous for fly ash and silica fume.
- Hence, in the case of the AC and given that the physical state of its hydraulic factors is totally amorphous, said influence of the pozzolan hydration on the OPC is TOTAL and, furthermore, from the very moment of its joint hydration beginning [17-29,32-48] with the added paradox that the first of both that has to be hydrated is the OPC to generate enough portlandite that the AC needs to promote, initiate and develop its pozzolanic reaction and thus end up being the one that continues and directs, from its beginning, the hydration of both at the same time but at the hydration rate of the AC (which is very fast [17-29,32-34]), but not that of the C3A of the pure OPC (which is much less fast [17-29]), being estimated ≈ 6 times smaller because, according to Von Weimarn’s and Garrido’s set patterns [99,100], the size of its slow-forming ettringite crystals must be and is ≈ 6 times larger [20-25,29]), until its original Al2O3r– (%) content is exhausted, and that of SiO2r– (%) content much later, which is much less significant for the initial purpose of the rapid hydration of its blended cement. Hence, AC has such a considerable influence on the rapid hydration of the OPC fraction with which it is mixed, from the beginning to the end of the pozzolanic activity of its Al2O3r– (%) content mostly and, above all. Both in the fresh state of its pastes with PC (rheological and thixotropic behaviours [47,48]), and in its newly set and initial hardening state (calorimetric behaviour [42,46] that influences the plastic behaviour, thermal and shrinkage due to drying of its pastes) and in its completely hardened state: SAI [37-39,87-90] and durability already described on Section 2, last paragraph.
The reason for this TOTAL chemical influence of the AC pozzolanic reaction on the OPC hydration, with which it is mixed, lies in the stimulation of hydration via the direct route [72-74], non-direct [75] and indirect especially and above all [17-29,41-46] that AC (of its original Al2O3r– (%) content very especially and above all until it is exhausted) provokes to the OPC (to its C3A content (%) especially). The foregoing constitutes further evidence that the pozzolanic activity of the Al2O3r– in the AC is more specific than generic, in order for the C3A (%) content in the PC fraction with which they are blended to hydrate more and more quickly than C3S (%) content [17-29,42-48]. By contrast, SiO2r– activity must be considerably less specific or unspecific or anti-specific for the same result as hydration progresses [17,29-31,42,46] [17]. Which, in any case, will shorten, very significantly, the initial and final setting and hardening times of your pastes [43-45] to the point of even having to correct upwards the setting regulator amount in the resulting blended cement [17,19] which, as it is a natural gypsum stone that is so abundant in Spain, it will reduce the final economic cost of its manufacture.
However, if the physical state of the pozzolana is almost completely vitreous like that of FA, the participation, intervention and similar pozzolanic reaction of the corresponding reactive fractions (present in their more or less spherical particles) is quite delayed when to first dissolve the glass of its spheres that contained them. So, by then, the construction material containing them may have set and hardened a lot. Especially if, for them to be completely dissolved, they also require a very considerable additional regulated and mandatory water curing [88] because their mixing water is insufficient. And with chemical admixture that reduces mixing water, even more insufficient. Which, without a doubt, improves its hardening without perhaps having yet been able to dissolve the FA in its entirety so that its aforementioned reactive fractions can begin to act pozzolanically like those of the AC, but, ultimately, by then it will be too late, with which, under the same circumstances and everything else being equal, its influence on the rheological behaviour of its fresh pastes with PC [47], in its newly set and hardened [43,65,66] and in the completely hardened [37,38] is considerably less than that of the ACs. Which, however, and in any case, does not justify that they do not have to be ground separately because in addition and usually their degree of hardness is higher than that of Portland clinker. That is, the opposite of what happens with AC as they are the result of geochemical and metamorphic reactions of the feldspars from granites origin mostly.
In short, the chemical justification of separate grinding (plus a Blending station) necessarily implies the physical justification although, in any case, both, physical and chemical justification, are subject to whether the cement Plant has necessary CAPEX for its implementation, in which case, it will be necessary to use some other more economical method if the quality of the matrix clay allows it, and the cement Plant and/or concrete Plant allows it as well, although always to the detriment of the technical and economic benefits referred to in the first 5 paragraphs of this Section. Everything stated about the chemical justification of separate grinding and the Blending station also justifies, in turn, Conclusion 4.
CONCLUSIONS
- Without a doubt, AC is beginning to establish themselves as the SCM with the greatest potential to replace Portland cement. These are artificial pozzolans, siliceous and aluminous in nature, according to EN 197-1:2011 [14] and ASTM C618-19 Standards [16], but aluminic in chemical character, according to R. Talero [17,29,31-47], of high to very high quality, depending on their reactive chemical composition (SiO2r– (%) content and Al2O3r– (%) content chiefly) controllable in their manufacture and sustainable with the environment, reducing CO2 emissions in their production by up to 75%, compared to clinker Portland according to its quality: high Al2O3r– (%) content.
- Clays that can be thermally activated are not only kaolinitic but also illitic and montmorillonitic (smectic). But, in any case, its feasibility of use and percentage of replacement by Portland cement depends fundamentally on its Al2O3r– (%) content first and foremost, that we can generate it during the process of their thermal activation on an industrial scale in cement Plants and/or concrete Plants.
- The process control of its thermal activation, in terms of maintaining the optimal calcination temperature in the kiln, the aerobic or anaerobic conditions and the speed of transit of the raw clay through it, is essential to guarantee the greatest pozzolanic activity of the AC produced. It is for this reason that the permanent control of the process variables and, among all of them, the Al2O3r– (%) content parameter of the newly manufactured AC, more than that of SiO2r– (%), is of utmost importance to correlate them:
- with the other chemical parameters (LOI, residual water and pozzolanicity in particular), which have been determined it before, and mechanical parameters (SAI), at the same time or after, all of them derived from its reactive chemical parameter: Al2O3r– (%) content first and foremost, but not the other way around, and
- with the other performance variables in the construction reality.
- The separate grinding and blending station are the best grinding option in terms of quality, granulometric distribution, grinding efficiency and economic cost. This conclusion had been deduced, chemically justified, and made public by R. Talero3 in 1986, its foundation appearing written in the text of the MEMORY of his PhD Thesis [17], but, above all, in the penultimate paragraph of its 10th Application, point 5, which in a highly summarized and synthesized manner reads as follows: "In presence of active mineral additions, separate grinding is predominant" (and non-active too, logically). Its basis and chemical justification, see the last 7 paragraphs of Section 7.
- Finally, and in any case, the most important thing of all is that Portland/AC cements are much more eco-efficient and environmentally friendly than the current ones, as they can minimize the associated and subsidiary generation of CO2 in cement manufacturing considerably more, the higher the Al2O3r– (%) content of their AC, by allowing higher percentages of replacement by OPC and, ultimately, much more sustainable than the current ones, in addition to reporting better mechanical, economic and durable performance against the attack of: chlorides on the steel reinforcements of concrete, sulphates (in replacement amounts by PC greater than 40%, 50% and even 55% or even more the higher its Al₂O₃r− (%) content than not, instead of SiO₂r− (%)), seawater and alkali-silica reaction (ASR), among other advantages.
NOTES:
- The remaining sedimentary materials, which can also be activated like clays, and therefore equally or even more usable as SCM, will be the subject from another communication.
- This communication has been prepared considering EU cement regulations [14], where the following replacements are authorized:
- In cement Plants, Portland clinker can be replaced by SCM type GGBFS (S); SF (D), NP (natural, P, or calcined, Q – this type includes AC), FA (siliceous, V, and calcareous, W), calcined shale (T), and limestone filler (L and LL); and
- In ready-mix concrete Plants [88,89], OPC CEM I type can be replaced by FA V type and SF (D).
In contrast, in other industrialized countries, only the latter option is authorized, although it is extended to many other SCMs, including AC. This significantly simplifies their manufacture, storage, and batching once they have been accepted due to positive quality control and its verification. However, such manufacturing is much more feasible for a group of ready-mix concrete Plants than for a single Plant, naturally.
RECOMMENDATION
As it has been seen, this communication lays the foundations and provides the technical means to be able to manufacture AC and quickly verify their quality in conventional cement Plants, using a simple, accurate, fast and economical wet analytical chemical method [50]. Although it is highly recommended not to manufacture them at the same time as the Portland clinker, but alternatively and with enough time to ensure that the air around the cement Plant does not reach high levels of the majority reaction products resulting from the dehydroxylation of the AC (H2O) and the decarbonation of the limestone in the clinker crude (CO2). Since the solubility of the latter in water can be quite considerable [101,102], but of all of it and, in any case, ≈ 1% is in the form of carbonic acid (H2CO3) and the rest in the form of dissolved gaseous CO2 [101] which is also very aggressive for the environment as well. Logically, this recommendation is not applicable to a group of ready-mix concrete Plants that manufacture AC.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this communication.
REFERENCES
- Redondo-Soto C, Morales-Cantero A, Cuesta A, Santacruz I, Gastaldi C, García-Aranda MA. (2023). Calcined clays for low carbon cement: Rheological behaviour in fresh Portland cement pastes. Cement and Concrete Research. 163:107018.
- Xuerun Li., Dengler J, Hesse C. (2023). Reducing clinker factor in limestone calcined clay-slag cement using C-S-H seeding – A way towards sustainable binder. Cement and Concrete Research.168:107151.
- Rathnarajan S, Dhanya BS, Pillai R, Gettu R, Santhanam M. (2022). Carbonation model for concretes with fly ash, slag, and limestone calcined clay - using accelerated and five - year natural exposure data. Cement and Concrete Composites 126:104329.
- Bhattacherjee S, Jain S, Santhanam M. (2022). A method to increase the workability retention of concrete with limestone calcined clay based cementitious system using a dispersing agent containing sodium hexametaphosphate. Cement and Concrete Composites. 132:104624.
- EN 196-5:2006 Standard. Methods of testing cement. Part 5. Pozzolanicity test for pozzolanic cements. AENOR. Calle Génova 6, 28040 Madrid – Spain.
- Talero R, Martín-Luengo M.P. (1992). Behaviour of grinded brick used as mineral admixture to Portland cements against gypsum attack. II Jornada sobre Aplicaciones Arquitectónicas de los Materiales Compuestos y Aditivados, ACTAS, p.83-102, ETS Arquitectura de Madrid-UPM.
- Martin-Luengo MP. Contribución al análisis y estudio del comportamiento en resdistencia sulfática y mecánicas de algunos componentes cristalinos y amorfos de las cenizas volantes. ThD Thesis. UAM – España, 12 junio 1997.
- Baroni G, Binda L. (1997). Study of the pozzolanicity of some bricks and clays. Construction and Building Materials. 11(1):41-46.
- Ngoc-Tra Lam M, Nguyen DT, Nguyen DL. (2021). Potential use of clay brick waste powder and ceramic waste aggregate in mortar. Construction and Building Materials. 313:125516.
- Navratilova E, Rovnaníkova P. (2016). Pozzolanic properties of brick powders and their effect on the properties of modified lime mortars. Construction and Building Materials. 120:530-539.
- Schackow A, Stringari D, Senff L, Correia SL, Segadaes AM. (2015). Influence of fired clay brick waste additions on the durability of mortars. Cement and Concrete Composites. 62:82–89.
- Grimshaw RW. (1971) The Chemistry and Physics of CLAYS. 4th Edition Revised. Ernest Benn Limited. London UK.
- Trusilewicz L, Fernández-Martínez F, Rahhal V, Talero R. (2012). TEM and SAED Characterization of Metakaolin, Pozzolanic Activity. Journal of the American Ceramic Society. 95(9):2989-2996.
- EN 197-1:2011 Standard. Cement. Part 1: Composition, specifications y conformity criteria for common cements. AENOR.
- Instrucción para la Recepción de Cementos RC-16. R.D. 256/2016 de 10 de junio.
- ASTM C 618-19 Standard. Standard Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use in Concrete. Annual Book of ASTM Standards, Section 4 Construction, Vol. 04.02 Concrete and Aggregates.
- Talero R. (1986). Contribución al estudio analítico y fisicoquímico del sistema: Cementos Puzolánicos-Yeso-Agua (a 20 ± 2 °C). PhD Thesis. UCM – España, 20 Nov.
- Talero R. (1996). Comparative XRD analysis ettringite originating from pozzolan and from Portland cement. Cement and Concrete Research. 26(8):1277-1283.
- Talero R, Bollati MR, Hernández-Olivares F. (1999). Manufacturing non-traditional mortars and concretes by OPC, metakaolin and gypsum (15,05 %). Materiales de Construcción. 49(256):29-41.
- Talero R. (2002). Kinetochemical and morphological differentiation of ettringites by Le Chatelier-Ansttet test. Cement and Concrete Research. 32 (5):707-717.
- Talero R. (2003). Kinetochemical and Morphological Differentiation of Ettringite by Metakaolin, Portland Cements and the Le Chatelier-Ansttet Test, Parameter: Vicat Needle Penetration vs. Time. Silicates Industriels. 68(11-12):137-146.
- Talero R. (2005). Performance of metakaolin and Portland cement in ettringite formation as determined by ASTM C 452-68: Kinetic and morphological differences. Cement and Concrete Research. 35(7):1269-1284.
- Talero R. (2007). Performance of metakaolin and Portland cements in ettringite formation as determined by Le Chatelier-Ansttet test: Kinetic and morphological differences and new specification. Silicates Industriels. 72(11-12):191–204.
- Talero R. (2008). Kinetic and morphological differentiation of ettringites by metakaolin, Portland cements and ASTM C 425-68 test, Part I: Kinetic differentiation. Materiales de Construcción. 58 (292):45-66.
- Talero R. (2009). Kinetic and morphological differentiation of ettringites by metakaolin, Portland cements and ASTM C 425-68 test, Part II: Morphological differentiation y SEM and XRD analysis. Materiales de Construcción. 59 (293):35-51.
- Rahhal V, Talero R. (2014). Very early detection of ettringite from pozzolan origin. Construction and Building Materials. 53:674-679.
- Talero R. (2010). Expansive synergic effect of ettringite from pozzolan (metakaolin) and from OPC, co-precipitating in a common plaster-bearing solution, Part I: by cement pastes and mortars. Construction and Building Materials. 24:1779-1789.
- Talero R. (2011). Co-precipitation of fast– and slow–forming ettringite. Consequence: Expansive Synergic Effect. Its demonstration using mortars and concrete. Materiales de Construcción. 61(303):327-352.
- Talero R. (2011). Expansive synergic effect of ettringite from pozzolan (metakaolin) and from OPC, co-precipitating in a common plaster-bearing solution, Part II: Fundamentals, explanation, and justification. Construction and Building Materials. 25:1139-1158.
- Talero R. (2012). Gypsum attack: performance of silicic pozzolans and Portland cements as determined by ASTM C 452-68. Advances in Cement Research. 24(1):1-15.
- Talero R. (2013). Performance of silicic pozzolans and Portland cements in their behaviour in front of gypsum attack as determined by ASTM C 452-68. Materiales de Construcción. 63 (310), 159-193.
- Arámburo C. (2019). Comportamiento sulfático y mecánico-resistente de cementos portland con elevados contenidos de puzolanas (> 40%): su fundamento químico-físico y justificación de otras consecuencias posibles. PhD Thesis. UPM – España, 12 junio.
- Arámburo C, Pedrajas C, Talero R. (2020). Portland cements with high content of calcined clay: mechanical strength behaviour and sulphate durability. Materials. 13 (18):4206.
- Pedrajas C, Arámburo C, Talero R. (2023). Sulphate durability of low carbon cements with high contents of calcined clay. Reasons for the resistant phenomenon. Materials Today: Proceedings. 1-6.
- Lannegrand R, Ramos G, Talero R. (2001). Condition of knowledge about the Friedel's salt. Materiales de Construcción. 51(262):63-71.
- Jones MR, Macphee DE, Chudek JA, Hunter G, Lannegrand R, Talero R. (2003). Scrimeneour S. N. Studies using 27Al MAS RMN of AFm and AFt phases and the formation of Friedel's salt. Cement and Concrete Research. 33:177-182.
- Mejía R. Contribución al estudio analítico y fisicoquímico del sistema: cementos Portland, puzolanas, escoria siderúrgica, cloruros y agua (a 20 ± 2 °C). PhD Thesis. UCM – España, 29 junio 1997.
- Mejía R, Delvasto S, Talero R. (2000). A new pozzolan high performance cementitious material. Materiales de Construcción. 50(260):5-13.
- Mejía R, Delvasto S, Talero R. (2003). Chloride diffusion measured by a modified permeability test in normal and blended cements. Adv Cem Res. 15(3):113-118.
- Talero R, et al. (2011) Comparative and semi-quantitative XRD analysis of Friedel’s salt originating from pozzolan and Portland cement. Construction and Building Materials. 25(5):2370−2380.
- Talero R, Trusilewicz L. (2012). Morphological Differentiation and Crystal Growth Form of Friedel’s Salt Originated from Pozzolan and Portland Cement. Industrial & Engineering Chemistry Research. 51:12517−12529.
- Talero R. (2012). Synergic effect of Friedel’s salt from pozzolan and from OPC co-precipitating in a chloride solution. Construction and Building Materials. 33:164-180.
- Rahhal V. (2002). Caracterización de adiciones puzolánicas por calorimetría de conducción. PhD Thesis, Dpto. Ingeniería Civil-Construcción-UPM – España, 12 dic.
- Talero R, Rahhal V. (2007). Influence of aluminic pozzolans, quartz and gypsum additions on Portland cement hydration. 12th Intern. Congress on the Chemistry of Cement, Proceedings, Montreal, Canada, 8-13 July.
- Rahhal V, Talero R. (2008). Calorimetry of Portland Cement with Metakaolins, Quartz and Gypsum Additions. Journal of Thermal Analysis and Calorimetry. 91(3):825-834.
- Talero R, Rahhal V. (2009). Calorimetric comparison of Portland cement containing silica fume and metakaolin: Is silica fume, like metakaolin, characterized by pozzolanic activity that is more specific than generic? Journal of Thermal Analysis and Calorimetry. 96(2):383-393.
- Pedrajas C. (2016). Contribución al análisis y estudio del comportamiento reológico de pastas frescas de cemento portland con adiciones minerales activas, tipo puzolana, y no activas, tipo filler PhD Thesis, UCLM – España, 8 feb.
- Arámburo C, Pedrajas C, Rahhal V, González M, Talero R. (2019). Calcined clays for low carbon cement: rheological behaviour in fresh Portland cement pastes. Materials Letters. 239:24-28.
- UNE 80225:2012 Standard. Methods of testing cement. Chemical analysis. Determination of reactive SiO2 content in cements, pozzolans and fly ash. AENOR.
- Talero R. (2017). Nuevo método de análisis químico por vía húmeda para determinar el contenido de alúmina reactiva de las puzolanas naturales y artificiales. PhD Thesis. UCM – España, 6 julio.
- Melgarejo JC (Editor). (2003). Atlas de asociaciones minerales en lámina delgada. Vol. I Caps. 12 (Los minerales de la arcilla. Autora: Monserrat Inglés) y 29 (Asociaciones minerales en procesos cerámicos. Autores: Álvarez-Pérez A, Prada-Pérez JL.) Fundación Folch. Univ. de Barcelona – España. 1ª Edición: UB 1997, Reedición: Publicaciones.
- Grim RF. (1968). Clay Mineralogy. Mc Graw-Hill, Nueva York 596.
- Bailey SW. (1980). Structures of layer silicates. En: Brindley, G.W., Brown G. (eds.): Crystal structures of clay minerals and their X-ray identification. Soc. Monograph. 5, 1-123.
- Deer WA, Howie R, Zussman J. (1966). An introduction to the rock forming minerals. Longman Group Limited. London, UK.
- Walter Maresch, Olof Medenbach (colaboración: Hans Dieter Trochim). Rocas. Título original: GESTEINE. Traducción: Rosa Sala, Revisión: Dr. Solans. Univ. de Barcelona, © Mosaik Verlag, Munich, 1987, © Naturart, S.A. Barcelona 1990, ISBN 84-87535-21-6, Depósito legal: B-41381-1990. Impreso en Gayban Grafic, S.A. Barcelona.
- Thompson AB. (1976). Mineral reactions in pelitic rocks: 1, Prediction of P-T-X (Fe-Mg); phase relations, II, Calculations of some P-T-X (Fe-Mg); phase relations. American Journal of Science. 276:401-424.
- Symmes GH, Ferry JM. (1992). The effect of whole-rock MnO-content on the stability of garnet in pelitic schists during metamorphism. Journal of Metamorphic Geology. 10:221-237.
- Koons PO, Thompson AB. (1985). Non-mafic rocks in the greenschist, blueschist and eclogite facies. Chemical Geology. 50:3-30.
- Spear FS. (1993). Metamorphic phase equilibria and pressure-temperature-time paths. Mineralogical Society of America. Monographs. 824.
- Thompson JB, Thompson AB. (1976). A model system for mineral facies in pelitic schists. Contributions to Mineralogy and Petrology. 58:243-277.
- Stone M, Exley CS. (1985). HHP granites of SW England and their associated mineralization. A review. High Heat Production granites. IMM. 571-593, London.
- Mangas J. (1987). Estudio de las inclusiones fluidas en los yacimientos españoles de estaño ligados a granitos hercínicos. PhD Thesis, Universidad de Salamanca – Spain, 595 págs.
- ITGE. (1993). Mapa Metalográfico de España a escala 1/2000.000 núm. 18 (Ponferrada). ITGE. Madrid.
- Mackenzie RC. (1957). The Diferential Thermal Investigation of Clays. Edited By Robert C. Mackenzie. Mineralogical Society (Clay Moneralogical Group). London.
- Rahhal V, Talero R. (2004). Influence of two different fly ashes on the hydration of Portland cements. Journal of Thermal Analysis and Calorimetry. 78:191-205.
- Rahhal V, Talero R. (2009). Fast Physics-Chemical Characterization of Fly Ash. Journal of Thermal Analysis and Calorimetry. 96 (2):369-374.
- Avet F, Li X, Scrivener K. (2018). Determination of the amount of reacted metakaolin in calcined clay blends. Cement and Concrete Research. 106:40–48.
- Avet F, Scrivener K. (2017). Hydration Study of Limestone Calcined Clay Cement (LC3) Using Various Grades of Calcined Kaolinitic Clays, in: F. Martirena, Aurélie Favier, K. Scrivener (Eds.), Calcined Clays Sustain. Concr. Proc. 2nd Int. Conf. Calcined Clays Sustain. Concr., Springer-Verlag France pp. 35-41.
- Scrivener K, Martirena F, Bishnoi S, Maity S. (2018). Calcined clay limestone cements (LC3). Cement and Concrete Research. 114:49–56.
- Irassar EF, Rahhal V, Bonavetti L, Castellano C. (2019). Blended cement with illitic calcined clay and calcareous material. 15th 15th International Congress on the Chemistry of Cement. Communication No. 379. Prague, Czech Republic, Sept. 16–20
- Irassar EF, Córdoba G, Zito S, Rossetti A, Rahhal V, Dario F. (2019) Durability of blended cements containing illitic calcined clays. 15th 15th International Congress on the Chemistry of Cement. Communication No. 253. Prague, Czech Republic, Sept 16–2.
- Rahhal V, Talero R. (2003). Calorimetric influence of two crystalline additions on hydration of Portland cements. The Journal of the Argentine Chemical Society. 91(4):91-101.
- Rahhal V, Talero R. (2005). Early hydration of Portland cement with crystalline mineral additions. Cement and Concrete Research. 35:1285-1291.
- Rahhal V, Bonavetti V, Delgado A, Pedrajas, Talero R. (2009). Scheme of the Portland cement hydration with crystalline mineral admixtures and other aspects. Silicates Industriels. 74(11-12):347-52.
- Rahhal V, Bonavetti L, Trusilewicz L, Pedrajas C, Talero R. (2012). Role of the fíller on Portland cement hydration at early ages. Construction and Building Materials. 27:82-90.
- Tironi A, Trezza MA, Scian AN, Irassar EF. (2013). Assessment of pozzolanic activity of different calcined clays. Cement and Concrete Composites. 37:319-327.
- Holt Erika E. (2000). Where did these cracks come from? ACI Concrete International, Sept.
- Kennedy TB. (1961). Significance of test for calcium sulphate in hydrated Portland-cement mortar. Discussion. Committee Reports - Technical papers; Proceedings of the American Society for Testing and Materials. 61:10-351042.
- Powers TC. (1949). The Nonevaporable Water Content of Hardened Portland-Cement Paste: Its Significance for Concrete Research and Its Method of Determination. ASTM Bulletin. 68:68-76.
- Powers TC. (1961). Some Physical Aspects of the Hydration of Portland Cement. Journal of the Research and Development Laboratories. 47-56.
- Brouwers JH. (2004). The work of Powers and Brownyard revisited: Part I. Cement and Concrete Research. 34:1697-1716.
- Brouwers H.J.H. (2005) The work of Powers and Brownyard revisited: Part 2. Cement and Concrete Research, Vol. 35, pp. 1922-1936.
- Autogenous Shrinkage of Concrete. Proceedings of the international workshop organized by the JCI (1998) (Japan Concrete Institute), Hiroshima.
- Aïtcin PC, Neville AM, Acker P. (1997). Integrated view of shrinkage deformation. Concrete International.
- Morin R, Haddad G, Aitcin PC. (2002). Crack Free High Performance Concrete Structures, ACI Concrete International.
- Neville Adams: (1984) Tecnología del concreto, Tomo 2, IMCYC, México.
- Pliego de Condiciones para la Recepción de Conglomerantes Hidráulicos, PCCH-64 (O.M., 9 de abril de 1964, B.O.E. Núm. 109, 9 de mayo de 1964 y B.O.E. nº 222, 15 de septiembre de 1964.
- Código Estructural. Real Decreto 470/2021, de 29 de junio (BOE núm. 190, 10 de agosto 2021).
- EN 450-1:2013 Standard. Fly ash for concrete. Part 1. Definitions, specifications y conformity criteria. AENOR.
- ASTM C311-87 Standard. Standard Test Methods for Sampling and Testing Fly Ash or Natural Pozzolans for Use in Portland-Cement Concrete. 1987, ASTM C311-94b, (1995).
- Leco Empowering Results. RC612 multiphase determiner. Determination of carbon and water and residual and diffusible hydrogen.
- EN 197-5:2021 Standard. Cement. Part 5: Portland-composite cement CEM II/C-M and Composite cement CEM VI. AENOR.
- EN 15743:2010+A1 Standard. Supersulphated cement. Composition, specification and conformity criteria. Dic. 2015. AENOR.
- Gruskovnjak A, Lothenbach B, Winnefeld F, Münch B, Figi R, Suz-Chung Ko, Adler M, Mäder U. (2011) Quantification of hydration phases in supersulphated cements: review and new approaches. Advances in Cement Research. 23(6):265-275.
- ASTM C452-68 Standard. Standard Test Method for Potential Expansion of Portland Cement Mortars Exposed to Sulphate.
- EN 13282-1 Standard. Conglomerantes hidráulicos para carreteras. Parte. 1: Conglomerantes hidráulicos para carreteras de endurecimiento rápido. Composición, especificaciones y criterios de conformidad. Nov. 2013. AENOR.
- EN 13282-2 Standard. Conglomerantes hidráulicos para carreteras. Parte. 2: Conglomerantes hidráulicos para carreteras de endurecimiento lento. Composición, especificaciones y criterios de conformidad. Marzo 2016. AENOR.
- ASTM C1157 Standard. Standard Performance Specification for Blended Hydraulic Cement.
- Von Weimarn P. (1952). Principles and Methods of Chemical Analysis, Prentice Hall, New York, p. 21.
- Garrido L. (1973). FORMA Y ESTRUCTURA DE LOS CRISTALES. Ed. Alhambra, S.A., Printed in Spain; R.E.182, Calle Claudio Coello,76, Madrid - Spain, pp.76.
- Biczok I. (1972). La corrosión del hormigón y su protección. URMO, S.A. Ediciones, c/ Espartero, 10, Bilbao, España.
- Hurtado J.R. (1956) Química Y Microbiología Del Agua, Tip. VARGAS, S.A. – Caracas – Venezuela.
 Abstract
Abstract  PDF
PDF.png)