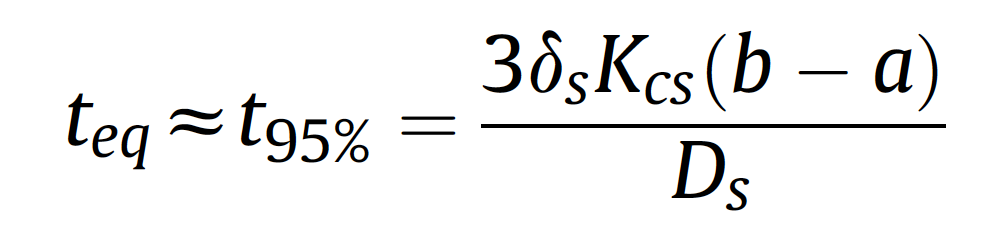Past Issues
Polybutylene Terephthalate/Ionic Liquid Nanofiber as a New Solid Phase Microextraction Coating
Ali Roostaie1*, Reza Haddad1, Sirwan Mohammadiazar2
1Institute of Police Equipment and Technologies, Policing Sciences and Social Studies Institute, Tehran, Iran
2Department of Chemistry, Islamic Azad University, Sanandaj Branch, Sanandaj, Iran
*Corresponding Author: Ali Roostaie, Institute of Police Equipment and Technologies, Policing Sciences and Social Studies Institute, Tehran, Iran. Tel: 9881886058, Email: [email protected]
Received: June 13, 2022
Published: September 28, 2022
Citation: Roostaie A, et al. (2022). Polybutylene Terephthalate/Ionic Liquid Nanofiber as a New Solid Phase Microextraction Coating. Material Science. 4(2):13.
Copyrights: Roostaie A, et al. © (2022).
ABSTRACT
In the present work, a new solid phase microextraction fiber coating based on ionic liquid doped polybutylene terephthalate (PBT) were prepared using electrospinning technique on unbreakable wire as a robust substrate. The synthesized PBT and modified PBT nanofibers were characterized by scanning electron microscopy. The results showed that modified PBT nanofibers have higher porosity and smaller diameter of nanofibers relative to non-modified PBT nanofibers. The feasibility of the method was considered using the isolation of selected chlorophenols as model compounds from water samples. The significant parameters of the electrospinning technique including the amount of ionic liquid, concentration of the polymer, electrospinning time and power voltage, and extraction parameters such as the extraction time and temperatures and the concentration of salt were studied and optimized. The developed method is sensitive and fast providing sub ng L-1 detection limits; the limit of detection of 0.75-5 ng L-1 and the limit of quantification of 5-15 ng L-1 with equilibrium time of 15 min were obtained. The linearity of the method was obtained in the range of 15-5000 ng L-1 with R2 >0.9997. The method repeatability at 100, 150, and 250 ng L-1 concentration level, RSD% of 4-10% was obtained. Finally, the proposed method was used to the analysis of real water samples and the relative recovery values were found to be in the range of 84-101%, showing no matrix effect.
Keywords: Modified polybutylene terephthalate, Ionic liquid, chlorophenols, Extraction, Aqueous sample.
INTRODUCTION
Solid phase microextraction (SPME) as a solventless and simple technique was developed in 1990 to perform preconcentration and isolation of the selected analytes in one step. In SPME method, firstly, the fiber coating of SPME device will be exposed to the sample headspace or immerse to the solution samples. Following definite time, which depends on the structure and thickness of the fiber coating and volatility of the sample, the equilibrium of the selected analytes between sorbent coating and sample is reached. In final stage, the extracted compounds are chemically or thermally desorbed and inserted to the analytical instruments for analysis [1]. In recent decades, the several geometries of SPME including unbreakable thin-film solid-phase microextraction (TF-SPME) [2] for simultaneous determination of the six most consumed artificial sweeteners in surface waters, Carbotrap B as a highly pure surface sorbent with excellent adsorption/dnderdesorption properties was packed into a stainless steel needle to develop a new needle trap device (NTD) [3] and copper tube which was served as both the substrate and sorbent with high physical strength and good flexibility [4] have been developed. Also, these have been focused on new developments in the use of sol-gel technology [5], molecularly-imprinted polymers (MIPs) [6] and electrochemical deposition [7] to prepare thermallystable, chemically-bonded and unbreakable SPME fibers.
One of the ways to improve the extraction efficiency of the SPME fiber coatings is to use electrospinning techniques to prepare nanocomposites and porous materials [8]. Electrospinning is a versatile and viable technique for generating ultrathin fibers. Remarkable progress has been made with regard to the development of electrospinning methods and engineering of electrospun nanofibers to suit or enable various applications [9]. The important parameter for uniform nanofibers without bead is the charge density of polymer solution [10] that depends on applied voltage [11] and ionic conductivity [12]. In recent times, ionic liquids (ILs) have been used to increase the ionic conductivity of the polymer solution and improve the morphology and structure of the nanofibers [13, 14]. The decrease of surface tension and viscosity of the monomer solution, are ideal to increase the aspect ratio of produced nanofibers consequently, extraction efficiency of coating will improve. Sun et al. [15] developed a fiber coated with a PIL-doped graphene oxide (GO). The monomers used were 1-(3-aminopropyl)-3-(4-vinylbenzyl) imidazolium 4-styrenesulfonate ([(APIM)(VB)] [SS]) and 1,6-di-(3-vinylimidazolium)hexane bis(hexafluorophosphate) ([C6 (VIM)2 2+] 2[PF6 ]) as a crosslinking agent. The prepared SPME fiber was applied to phenolic compound analyses in river water and groundwater of an industrial park. Luo et al. [16] studied the effect of solution viscosity behavior on the structure of the polybutylene succinate-co-terephthalates. Liu et al. [17], studied the rheological properties of 1-butyl-3- methylimidazolium chloride /polyacrylonitrile under different conditions. Diverse processing parameters including the polymer concentration, the solution flow rate, distance between the collector and needle, and the applied voltage are affected on the diameters of the fiber.
Here, we developed butyl-3-methylimidazolium chloridepolybutylene terephthalate nanocomposite as new SPME coating for the isolation and analysis of chlorophenols from aqueous samples. Our aim in this work is to improve mechanical strength of SPME method using unbreakable fiber while providing a sensitive and green technique. A polybutylene terephthalate containing butyl-3-methylimidazolium chloride (PBT–ILs) nanofibers was directly formed on a rotating stainless steel SPME needle, acting as an electrospinning collector. The addition of butyl-3-methylimidazolium chloride into polymer solution facilitated the electrospinning process that led to better elongation of electrospun nanofibers, reduction of the nanofibers diameters, and enhancement of their aspect ratio.
EXPERIMENTAL
Material and reagents
Selected target compounds were: phenol (Ph), 4-chlorophenol (4CP), 2,4-dichlorophenol (24DCP), and 2,4,6-trichlorophenol (246TCP) and they were provided from Merck (Darmstadt, Germany). The stock solution of chlorophenols was prepared in HPLC-grade methanol at a concentration of 1000 µg mL-1. Daily, the work solutions were freshly prepared by diluting the stock solution with methanol solvent. Polybutylene terephthalate (PBT) was purchased from Kolon Industries Inc. (Seoul, Korea) and triflouroaceticacid (TFA) were obtained from Riedel-de Haen (Germany).
Instrumentation For calibration and evaluation of current method, HP 6890 series gas chromatography (Hewlett −Packard, USA) connected to a HP 5973 mass − selective detector system (Varian Inc.) was used for GC/MS analysis. The separation of extracted compounds was performed by a column HP−5 ms (30 m, 0.25 mm i.d.) with 0.25 μm stationary phase. The column temperature was programmed at 55 °C for 3 min during desorption process, and was then increased to 180 °C with 15 °C min-1 and further to 260 °C with 20 °C min-1 (hold for 1 min). The SPME fibers were desorbed in a split/splitless injector, set to 200 °C and with the split initially closed for 3 min. For quantitative analysis, the MS was set on the selected ion monitoring (SIM) mode and two characteristics ions for each compound were monitored (Table 1). The scanning electron microscopy (SEM) images were recorded using Cambridge Stereoscan 360 SEM Instrument (England) operating at 20 kV.
Table 1: Retention times and selected ions of compounds studied by GC–MS.
|
Compounds |
Selected ions (m/z) |
|
Ph |
94, 136 |
|
4CP |
128, 170 |
|
24DCP |
162, 168 |
|
26DCP |
196, 198 |
Synthesis of 1-butyl-3-methylimidazolium chloride
The same molar of 1-chlorobuthan and 1-methylimidazolium were added into a round-bottomed flask fitted with a reflux condenser for about 48 h at 70o C with stirring until two phases formed. The top phase layer containing the unreacted starting materials was decanted and removed and then, ethyl acetate was added under mixing. The ethyl acetate was then decanted and the process was repeated two more times to remove the unreacted materials.
PBT/ILs nanocomposite preparation by electrospinning
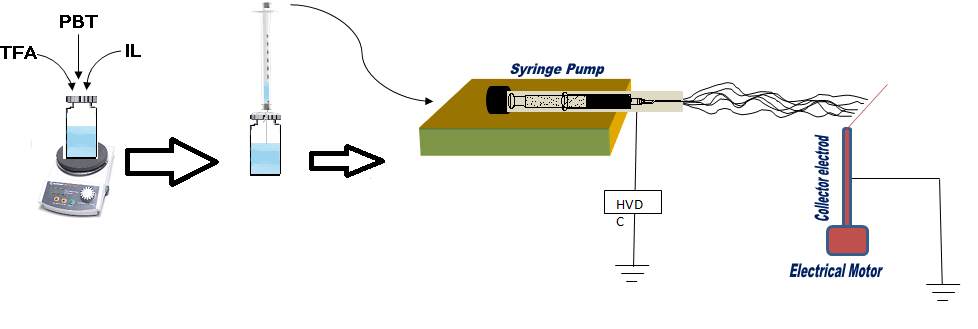
The PBT polymer containing 1-butyl-3-methylimidazolium chloride was prepared by home-made electrospinning apparatus. In the beginning, 14% (w/v) of PBT was added into 1 mL of TFA and stirred for 10 min to obtain homogeneous solution. Then, the different amounts of 1-butyl-3- methylimidazolium chloride were stirred for 10 min to obtain homogeneous solution. After that, the final solution was loaded into a syringe mounted in a syringe pump to deliver the solution at the flow rate of 0.5 μL min−1. The high voltage (15 kV) was applied between the collector (SPME substrate) and syringe needle containing polymer with the distance of 10 cm (Fig. 1). All the electrospinning processes were performed under ventilation.
Figure 1: The schematic of setup for synthesize the PBT/IL nanocomposite.
Headspace solid phase micro extraction analysis
The headspace solid phase microextraction (HS-SPME) of chlorophenols was carried by a manual home-made SPME holder containing the PBT/ILs nanocomposite coating. In all experiments, 0.8 g of sodium chloride was added to a 4 mL aqueous sample in a 10 mL vial containing a magnetic PTFE coated stir bar. The extraction temperature was set at 35 ◦C by a circulating water bath. After 15 min, the SPME fiber was withdrawn into the holder and inserted into the GC injection port for thermal desorption.
RESULTS AND DISCUSSION
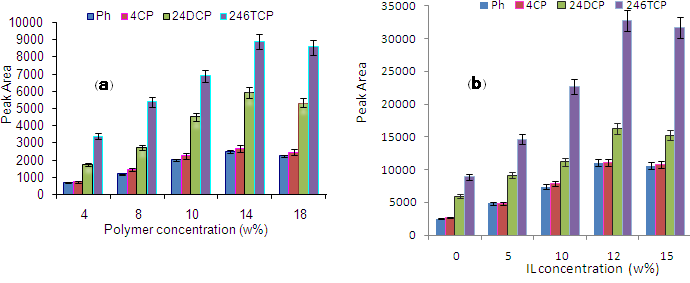
Owing to a number of PBT unique properties [18] such as (i) stiffness and high strength, (ii) high chemical resistance to different solvents and (iii) good wear characteristics and low water absorption, it efficiently is applied in numerous fields. The nanocompsites based on PBT have hydrophobic interactions, since they efficiently enable to remove aromatic organic compounds from aqueous sample. In this work, parameters influencing on the morphology and structure of the PBT/ILs nanocomposite including the concentration of polymer (4-18% (w/v)), the time of electrospinning and amount of ILs were investigated. As shown in Fig. 2a, by increasing the polymer concentration, the efficiency of extraction was enhanced until it reached to 14% and then, remained almost constant. Effect of ILs doping level on the efficiency of extraction was studied in the range of 0-20%. According to Fig. 2b, increasing the ILs doping level up to 12 % leads to an improvement in the extraction efficiency. At doping levels higher than 12 %, the electrospinning process became difficult to perform. Therefore, amount of 12 % was chosen as the optimum value. The time of electrospinning as another effective parameter on the coating thickness of SPME fiber was studied. When the time of electrospinning was applied longer, the thickness of coating becomes thicker and providing higher extraction efficiency. However, increasing the thickness of coating is cause to the longer the time of equilibrium. Therefore, the time of electrospinning of 15 min was selected as the time of equilibrium.
Figure 2: Effects of concentration of polymer (a) and amount of ionic liquid (b) on the extraction efficiency.
Characterization of the PBT/ILs
The SEM images of PBT/ILs nanocomposite are shown in Figure 3, and the porosity of the prepared coating with the diameter of nanofibers lower than 175 nm were confirmed. The SEM images of the nanofibres (Fig. 3) show that the addition of IL to the PBT polymer solution caused to diameter of nanofibers is decreased. The result is surely shown that the PBT/IL nanofibres have more pores compared to non-modified PBT, facilitating the mass transfer of the analytes during extraction/desorption process. Also, the narrower nanofibers increased the surface area of sorbent and available adsorption sites and therefore the efficiency of extraction. The addition of butyl-3-methylimidazolium chloride increased the ionic conductivity of the solution and caused reduction in surface tension of polymer solution. The whipping jet was more stretched in a solution with the less surface tension resulting in smaller fiber diameters.
Figure 3: SEM images of PBT nanofiber (a), PBT/IL nanocomposite at different scales (b) and (c).
Optimization of extraction conditions
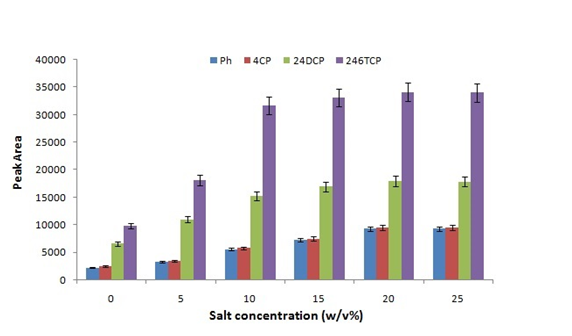
Influencing factors such as the time and temperatures of extraction, the concentration of salt and desorption process were studied. The increasing of the ionic strength of the aqueous solution would affect the solubility of organic solutes. The ionic strength was investigated in the range of 0–30% (w/v) of NaCl (Fig. 4a) and increase in peak area was observed up to 20% and then it remained constant. Therefore, further experiments were performed by the addition of 20% (w/v) NaCl.
Figure 4: Effects of ionic strength on the extraction efficiency.
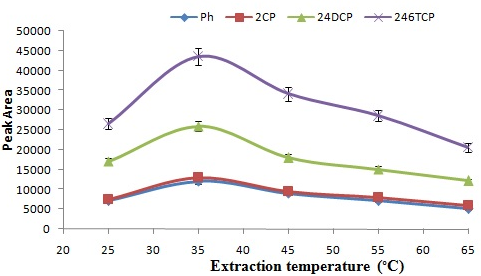
In headspace mode of SPME, the temperature of solution is a significant parameter and its effect on the efficiency of extraction was considered from 25 to 65 °C (Fig. 5). The efficiency of extraction was improved by increasing the temperature of extraction up to 35 °C, as analytes moved the headspace from the sample. However, a decrease was observed for some compounds at temperature greater than 35 °C due to the exothermic nature of the extraction.
Figure 5: Effects of ionic strength on the extraction efficiency.
The extraction efficiency is related to the expose time of the fiber coating and the analytes in the headspace phase. Extraction time profile was obtained in the ranging of 5 to 30 min. The results showed that with the increase in extraction time from 5 to 15 min, the extraction efficiency increased. According to [1]:
The equilibrium time is directly related to the boundary layer, partition coefficient and thickness of the coating and inversely to diffusion of the analytes in the coating. Rapid equilibrium of analytes between coating and the sample is related to the porous structure of the coating and large distances within the nanofibers (SEM images, Figure 3).
Performance evaluation of the fiber
The current method was evaluated in terms of repeatability, linearity, accuracy, limits of quantification and detection. The relative standard deviations for all analytes at a level of 100, 150, and 200 ng L−1 were obtained in the range of 4-10%. The method linearity was obtained at several spiked levels from 15 to 5000 ng L−1 and correlation coefficients (R) from 0.9997 to 0.9999 were obtained (Table 2). The limit of detection (LOD) and limit of quantification values were in the range of 0.75-5 ng L−1and 5-15 ng L−1, respectively. In addition, recovery tests were carried out by the proposed method using environmental and tap water sample spiked the analytes at 100 ng L−1.
Table 2: Some analytical data obtained after HS-SPME of selected chlorophenols using the PBT/IL nanocomposite fiber and GC–MS.
|
Compound |
LDR (ng L-1) |
R |
LOD (ng L-1) |
RSD% (n = 6) |
RSD% (n=3) |
Relative recovery (%) |
||||||
|
|
|
|
|
100 ng L-1 |
150 ng L-1 |
250 ng L-1 |
fiber- to- fibre |
Calan Dam |
Tap water |
Rainwater |
||
|
Ph |
15-5000 |
0.9997 |
5 |
10 |
10 |
8 |
9 |
86 |
86 |
90 |
||
|
4CP |
15-5000 |
0.9997 |
5 |
9 |
8 |
7 |
7 |
85 |
84 |
87 |
||
|
24DCP |
15-5000 |
0.9999 |
1 |
6 |
5 |
5 |
5 |
96 |
94 |
99 |
||
|
246DCP |
5-5000 |
0.9999 |
0.75 |
5 |
5 |
4 |
7 |
97 |
96 |
101 |
||
Table 3: Comparing the current work with some other methods.
|
Method |
LOD (ngL-1) |
RSD (%) |
Ref. |
|
Polyamid/ethylene glycol nanofibers
|
0.8–25 |
2.1–9.0 |
[19] |
|
1-butyl-3-methylimidazolium hexafluorophosphate coating
|
8–26 |
3.4–7 |
[20] |
|
Periodic mesoporous organosilica/ionic liquids
|
5–70 |
5.6–7.5 |
[21] |
|
Current method |
0.75-5 |
6-12 |
- |
In this study, a SPME-GC/MS method based on PBT/IL nanocomposite fiber coating was developed for trace determination of selected chlorophenols from aqueous samples. The SEM images proved the lower diameter of the prepared nanofibers in the presence of ILs resulting in the improved sensitivity of the SPME device. Relative recoveries along with other analytical data confirmed the suitability of the synthesized nanocomposite as an appropriate SPME fiber coating. Given the porous structure of the electrospun PBT/IL nanocomposite coating, high specific surface area with increased activated sites was achieved. The unique properties of the developed coating resulted in a short equilibrium time while providing the low ng L-1 level of detection. The developed method based on the use of PBT/IL nanocomposite coating, is facile, simple, rapid and inexpensive to effectively apply for determination of chlorophenols with sufficient sensitivity and reproducibility.
REFERENCES
- Piri-Moghadam H, Alam MN, Pawliszyn J. (2017). Review of geometries and coating materials in solid phase microextraction: Opportunities, limitations, and future perspectives. Anal Chim Acta. 984:42-65.
- Cárdenas-Soracá DM, Singh V, Nazdrajić E, Vasiljević T, Grandy JJ, Pawliszyn J. (2020). Development of thin-film solid-phase microextraction coating and method for determination of artificial sweeteners in surface waters. Talanta 211:120714.
- Poormohammadi A, Bahrami A, Farhadian M, Shahna FG, Ghiasvand A. (2017). Development of Carbotrap B-packed needle trap device for determination of volatile organic compounds in air. J Chromatogr A. 1527:33-42.
- Sun M, Feng J, Bu Y, Luo C. (20175). Highly sensitive copper fiber-in-tube solid-phase microextraction for online selective analysis of polycyclic aromatic hydrocarbons coupled with high performance liquid chromatography. J Chromatogr A. 1408:41-48.
- A. Amiri. (2017). Solid-phase microextraction-based sol–gel technique. Tr. Anal. Chem. 75:57-74.
- Asiabi H, Yamini Y, Seidi S, Ghahramanifard F. (2016). Preparation and evaluation of a novel molecularly imprinted polymer coating for selective extraction of indomethacin from biological samples by electrochemically controlled in-tube solid phase microextraction. Anal. Chim. Acta. 913:76-85.
- Lian C, Feng X, Tian M, Tian Y, Zhang Y. (2022). Electrodeposition of zeolitic imidazolate framework coating on stainless steel wire for solid-phase microextraction of polycyclic aromatic hydrocarbons in water samples. Microchem J. 175:107146.
- Najarzadegan H, Roostaie A, Ehteshami S. (2018). Electrospun Polyethylene Terephthalate/Graphene Nanocomposite as a New Solid-Phase Microextraction Fiber Coating for Enhanced Determination of Organochlorine Compounds. Chromatographia. 8(10)1413-1420.
- Xue J, Wu T, Dai Y, Xia Y. (2019). Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications. Chemical reviews. 119(8):5298-5415.
- Stanger J, Tucker N, Kirwan K, Staiger MP. (2009). Effect of charge density on the taylor cone in electrospinning, Int J Modern Phys. B. 23:1956-1961.
- Fallahi D, Rafizadeh M, Mohammadi N, Vahidi B. (2008). Effect of applied voltage on jet electric current and flow rate in electrospinning of polyacrylonitrile solutions. Polymer International. 57:1363-1368.
- Sheoran N, Boland B, Thornton S, Bochinski JR, Clarke LI. Increasing ionic conductivity within thermoplastics via commercial additives results in a dramatic decrease in fiber diameter from melt electrospinning. Soft Matter. 17(41):9264-9279.
- WX Dong, X Chen, YM Zhang, CS Wang, HP Wang. (2010). Rheological behavior of polyacrylonitrile/ionic liquid solutions. Chin. Syn. Fiber Ind. 33:33.
- Zavgorodnya O, Shamshina JL, Bonner JR, Rogers RD. (2017). Electrospinning biopolymers from ionic liquids requires control of different solution properties than volatile organic solvents. ACS Sustainable Chem. Eng. 5:5512–5519.
- Sun M, Bu Y, Feng J, Luo C. (2016). Graphene oxide reinforced polymeric ionic liquid monolith solid-phase microextraction sorbent for high-performance liquid chromatography analysis of phenolic compounds in aqueous environmental samples. J Sep Sci. 39:375–382.
- Luo SL, Li FX, Yu JY. (2009). Experimental investigation of viscoelastic behavior of biodegradable poly(butylene succinate-co-terephthalate) (PBST) fibers. Int. Nonlin. Sci. Num. 10:1339–1343.
- Liu WW, Cheng LY, Zhang HY, Zhang YM, Wang HP. (2007). Rheological behaviours of polyacrylonitrile/1-butyl-3-methylimidazolium chlorideconcentrated solutions. Int J Mol Sci. 8:180–188.
- Bagheri H, Roostaie A. (2015). Polybutylene terephthalate-nickel oxide nanocomposite as a fiber coating. Anal Chim Acta. 863:20–28.
- Bagheri H, Najarzadekan H, Roostaie A. (2014). Electrospun polyamide-polyethylene glycol nanofibers for headspace solid-phase microextration. J Sep Sci. 37(4):1880-1886.
- Ho T, Chen C, Li Z, Yang TC, Lee M. (2012). Determination of chlorophenols in landfill leachate using headspace sampling with ionic liquid-coated solid-phase microextraction fibers combined with gas chromatography-mass spectrometry. Analytica Chimica Acta 712:72– 77.
- Mousavi KZ, Yamini Y, Karimi B, Khataei MM, Khorasani M, Seidi S, Ghaemmaghami M. (2021). Plugged bifunctional periodic mesoporous organosilica as a high-performance solid phase microextraction coating for improving extraction efficiency of chlorophenols in different matrices. Talanta. 235:122724.
 Abstract
Abstract  PDF
PDF